|
In each individual measurement, the signal is indeed a convolution of affinity and concentration. As is well-known from literature (see examples 11, 12), decoupling affinity and concentration requires a multi-point titration experiment. To address this challenge, we developed a research method to quickly estimate antibody affinity directly in patient samples and used these data for evaluating the affinity dependence of diagnostic immunoassays. When interpreting measured antibody levels, it is important to understand the degree to which affinity biases the reported values. Measurements of antibody concentration are inextricably linked to their affinity. Clearly, both immunoassay formats rely on the patient antibody's ability to bind antigen. In this format, the sample, microparticles, and labelled conjugate are all combined simultaneously, which allows the conjugate to fill antibody binding sites that remain free from interacting with microparticles. Instead of secondary antibody, the same recombinant spike protein, labelled with a signal generating group, is employed. Direct immunoassays also capture patient antibodies with spike protein-coated microparticles but utilise a different detection approach. 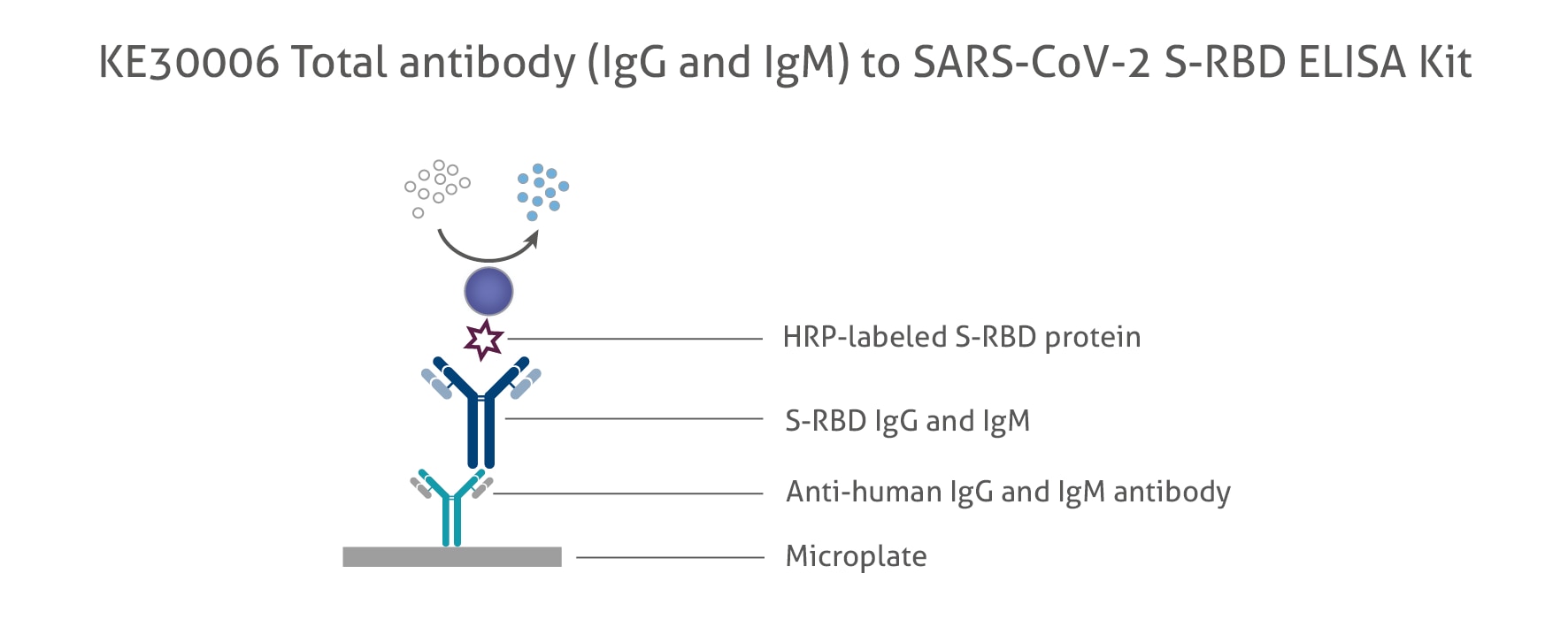
Indirect assays capture anti-spike antibodies from patient plasma/serum on microparticles coated with recombinant spike protein and detect them with labelled anti-human IgG or IgM secondary antibody (e.g., mouse anti-human). There are two common immunometric assay formats utilised for SARS-CoV-2 antibody detection on automated diagnostic platforms. 8, 9, 10įurthermore, the assay design also plays a role. The measured value reflects a convolution of patient antibody concentration and affinity, which both vary widely with time and the strength of the immune response. Therefore, quantifications of anti–SARS-CoV-2 antibody depend on both these parameters. For instance, a few antibodies that bind extremely well can create as many immune complexes as a higher concentration of poorly binding antibodies. Sandwich formation is driven by the affinity and concentration of the interacting molecules. 7 In these assays, the analyte is sandwiched between the capture reagent (to concentrate the target) and the detection reagent (to generate a signal). Immunometric (or sandwich) immunoassays have proven to be preeminent for measuring concentrations of diagnostically important macromolecules. 4, 5, 6 These quantitative tests, being developed under less urgent conditions, must be scrutinised for their ability to accurately assess the level of immune protection for tested individuals. 2, 3 Lately, the need has been increasingly shifting toward quantitative antibody measurements to monitor vaccine-acquired immune protection. Initially, SARS-CoV-2 qualitative antibody assays came into their own as an emergency means for determining seroprevalence. In particular, the ability to quickly screen a large number of patient samples is critical, especially for population surveillance, 1 which per force must come second to clinical needs in emergency conditions. The SARS-CoV-2 pandemic has underscored the need for quick and practical methods for assessing these parameters in COVID-19 patients and vaccinated individuals. Antibody affinity and concentration are fundamental characteristics of adaptive immunity against infections.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
